[ad_1]

Could a global policy shift be the best way to kick-start research into psilocybin’s medicinal potential?
A new movement for international psychedelic reform was started. A global coalition consisting of the Beckley Foundation, the Multidisciplinary Association For Psychedelic Studies (MAPS), Drug Science and Open Foundation and the agency Mind Medicine Australia launched this push last Tuesday.
Psychedelics have been banned in the US and internationally for several decades. This new decade seems to herald hopes for the decriminalization and medical use of psychedelics. The global coalition hopes to change international policy on banning the magic drug. The international shift of psilocybin mushrooms could embolden US lawmakers to follow suit.
Tired of foot dragging
Proponents have advocated countless times for legislation allowing the use of psychedelics to treat certain diseases. However, US lawmakers did a lot of foot-dragging instead. Finally, last year, a bill was passed that would allow limited research into the drug’s therapeutic potential.
The International Therapeutic Psilocybin Rescheduling Initiative (ITPRI) has begun its push for psychedelic reform. The panel has committed to reclassifying these substances, which have been banned under the United Nations 1971 Convention on Psychotropic Drugs. This 1971 directive makes psilocybin the most harmful drug on the list, hence the severe restriction on the psychedelic drug. In the US, psilocybin also fits into the Schedule 1 category. Many proponents of psychedelics and cannabis believe this classification should be reserved for “hard” drugs that pose serious health risks to people regardless of their region—drugs with little or no known medicinal benefits .
Recent developments have proven that psilocybin or magic mushrooms do not fit the criteria for Schedule I substances. The global organization pointed out that despite its medicinal potential, psilocybin is unfairly placed under strict limits. A key point raised during the congress is that psilocybin could be licensed, manufactured safely in limited quantities (for starters), and imported or exported based on acceptable regulations. This is better than the rough surveillance of the industry by international bodies.
A boost for more research
If this push is successful, Plan I researchers can study psilocybin, magic mushrooms, LSD, and other related psychedelics. To do this, these scientists also have to face up to international regulations. These hurdles don’t come cheap and add additional cost and complexity to research. The banning of the substance by governments around the world can negatively impact ethical approvals, collaborations and funding.
The first step in properly implementing this new measure will be to liaise with member states that are part of the signatories to initiate a formal review of their stance against psilocybin and other psychedelic substances. This review will bring to light the benefits and risks of the drug. To start the process, a country must apply for a review from the World Health Organization (WHO).
The WHO itself could also initiate the procedure. Once the application has been submitted and approved, the international organization is responsible. The Executive Committee on Drug Dependence (ECDD) would initiate the review process. Recommendations for policy changes are made by the member states. Any review that receives the support of two-thirds of signatory countries would be included in legislation, whether or not it included a recommendation for postponement
Christopher Kodderman, CEO and co-founder of ITPRI, said in a press release that a change in the status of psilocybin as a Schedule I drug is long overdue. He added that there is a better scientific understanding of the substance’s high therapeutic potential and low addictiveness. “That’s enough to turn the tables,” he explained.
Possible member states to initiate the procedure
ITPRI says it has identified about a dozen countries willing to propose a review to move psilocybin around the world. According to Mr. Koddermann, Canada is the most suitable option from the options available. Canada is one of the few countries to have fully decriminalized and legalized cannabis (a Schedule I substance). The country has also eased its restrictions on the use of entheogens in critically ill patients.
Founder of ITPRI partner Drug Science and head of Imperial College London’s Center for Psychedelic Research, David Butt, stressed the coalition’s shared view that existing international planning for psilocybin should support progress in developing treatments for neurological patients using the drug seriously restricted.
Willem Scholten, former WHO ECDD secretary and ITPRI advisory board member, added that the review of the 1971 convention guidelines is a game-changing opportunity to boost the psilocybin industry. This will accelerate the development of life-changing medicines for patients and explore other medicinal potentials of the drug.
The fears of the coalition
Just as one member country can initiate this review process, another can oppose it. Recently, the WHO maintained its stance against prescribing krotom (a plant known for its pain-relieving potential). This decision was made despite the results of a scientific review of the facility. ITPRI has concerns that WHO’s ECDD could do the same if a rescheduling recommendation for psilocybin is made. With kratom, the WHO could have proposed a more critical review, possibly resulting in a postponement, or persuaded member countries to regulate the drug.
In 2020, the United Nations Commission on Narcotic Drugs approved a WHO recommendation to move cannabis out of the most restrictive international classification category.
Psychedelics in the US
As the international coalition works tirelessly to advance the policy review initiative, pro-psychedelics groups in the United States are also working tirelessly to initiate reforms to existing federal policies regarding plants like psilocybin or magic mushrooms.
At the same time, bills are being introduced to the Senate and House of Representatives to end cannabis prohibition. Cannabis and psychedelics reforms are beginning to gain the attention of the Biden administration.
Rep. Earl Blumenauer has sought to work with the DEA to legalize the use of psilocybin by patients without fear of federal repercussions. He added that the drug would be used as an investigational treatment in ongoing clinical trials.
last
Millions of people around the world are excited by recent advances in psychedelics research. International and national policy changes will follow many of these incredible feats.
UN DRUG TREATIES, READ MORE..

DRUG UN TREAT BE DAMNED, GERMANY IMPORTS JAMAIC WEED!
OR..
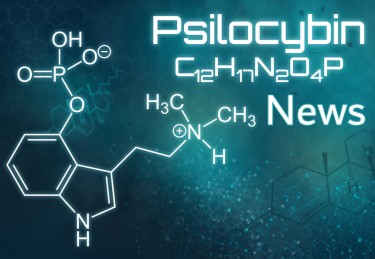
PSILOCYBIN NEWS – WHAT ARE THE LATEST HEADLINES?

